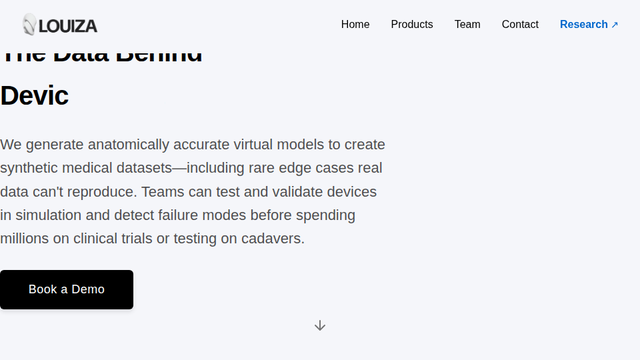
Simulative Biology | Louiza Labs
Simulative Biology | Louiza Labs
LOUIZA.AI
Created 03 Aug 2025
Brand Description Not Available
Industries
Medical Technology
Healthcare
Artificial Intelligence
Regulatory Compliance
Maggie Powers Every Post on the Fast Track to Growth
She refuels daily with SEO & GEO insights to better serve.
Unlock More Content Ideas
Create an account or log in to explore exclusive blog topics, SEO strategies, and GEO-targeted content generated by AI CMO Maggie
[ Tailored for your brand's next growth leap. ]
Maggie Studies the Tracks Before the Journey Begins.
She learns every detail of your business through deep market research.
Target Problem
Target Regions
Target Audience
Market Research
Maggie Maps Your Rivals Before Full Steam Ahead.
She benchmarks your brand against competitors to plot a smarter route.
Products & Services
Unique Selling Points
SWOT Analysis
Market Competitors
Launch Effortless Blogging with Maggie
No more blank pages. Maggie runs your blog with vibe-rich, SEO-tuned, GEO-smart content — built to be loved by search engines and surfaced by AI.

Free Tools
AI Ideas BrainstormingAI Startup Trend AnalysisAI Project Management......
AI Co-Founders
RoadmapAll rights reserved by AI Marketing OS Ltd. Designed & Developed by TOPY.AI .